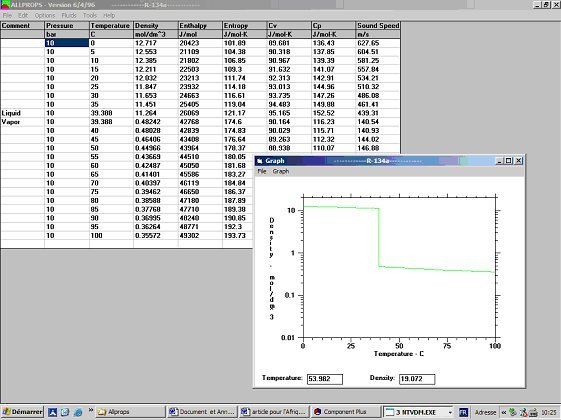
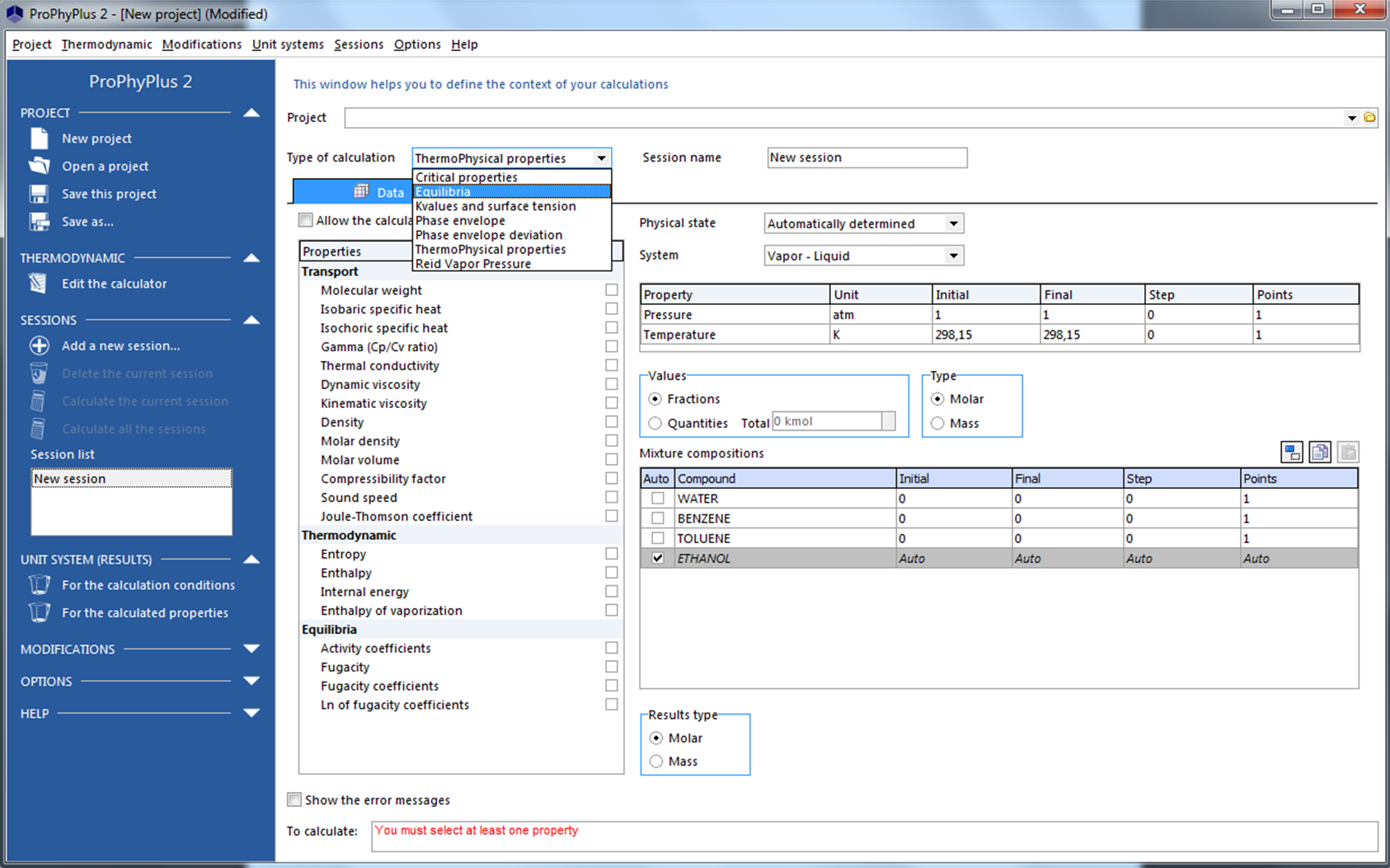
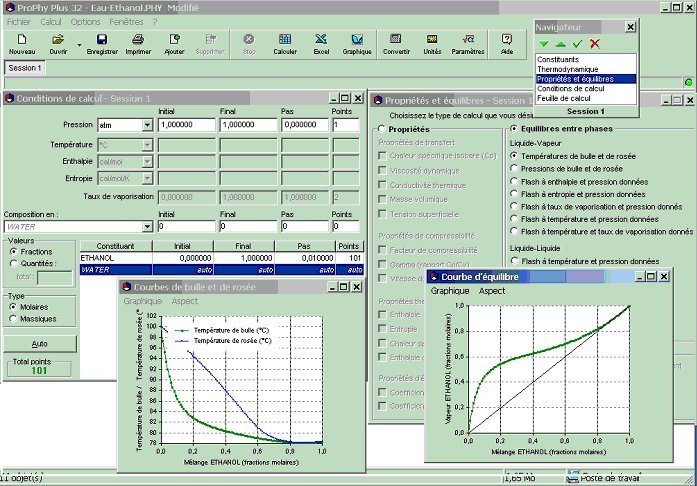
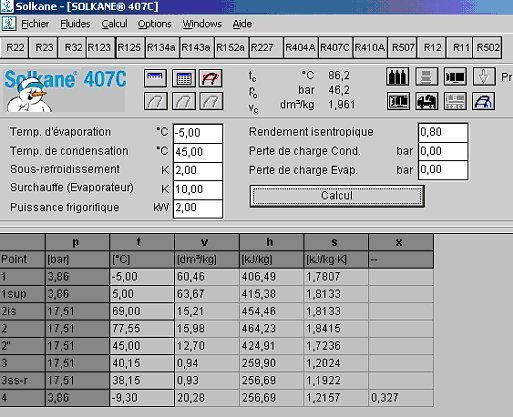
Refrigerant properties: R22 Properties, R22 tables, and R22 Data - Evaluate States and Properties of Refrigerant R22 (CClF3) Using TEST, a Web-based Application.
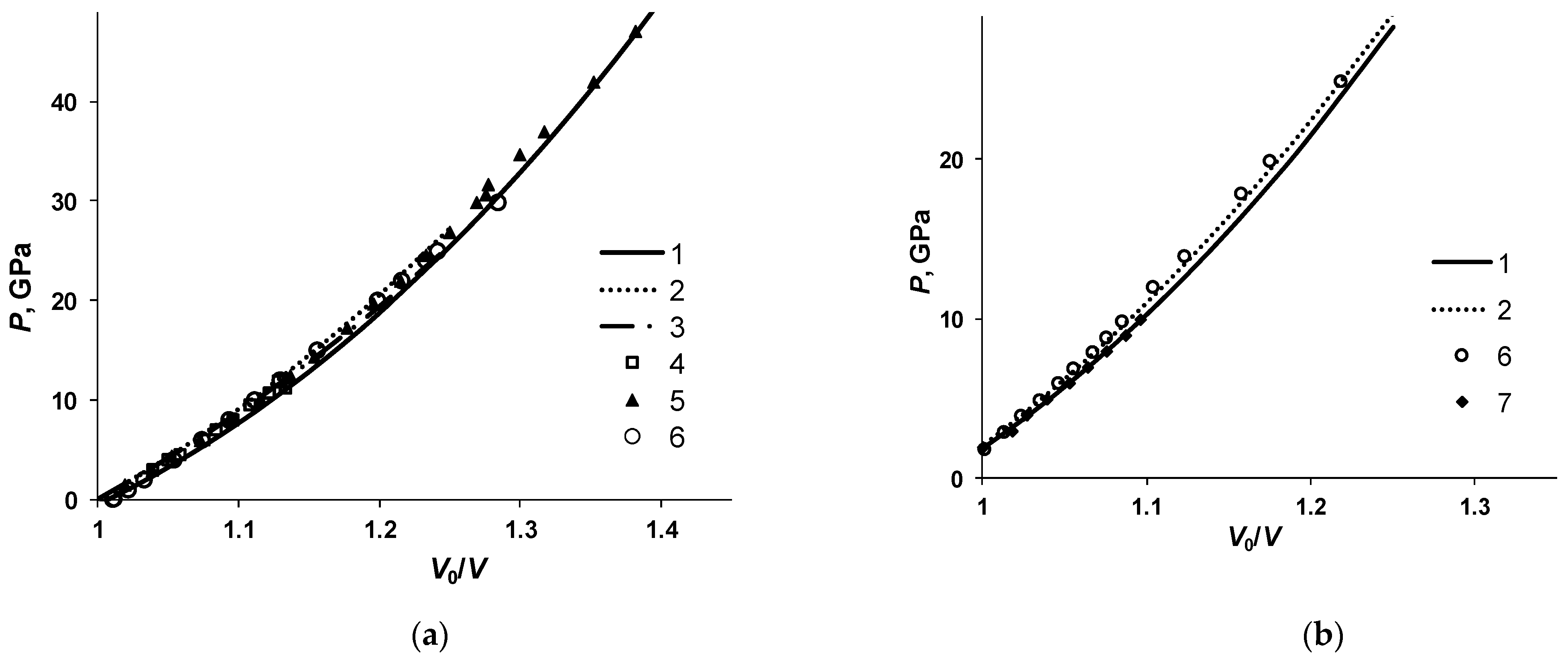
Metals | Free Full-Text | Calculation of Thermodynamic Properties of Metals and Their Binary Alloys by the Perturbation Theory



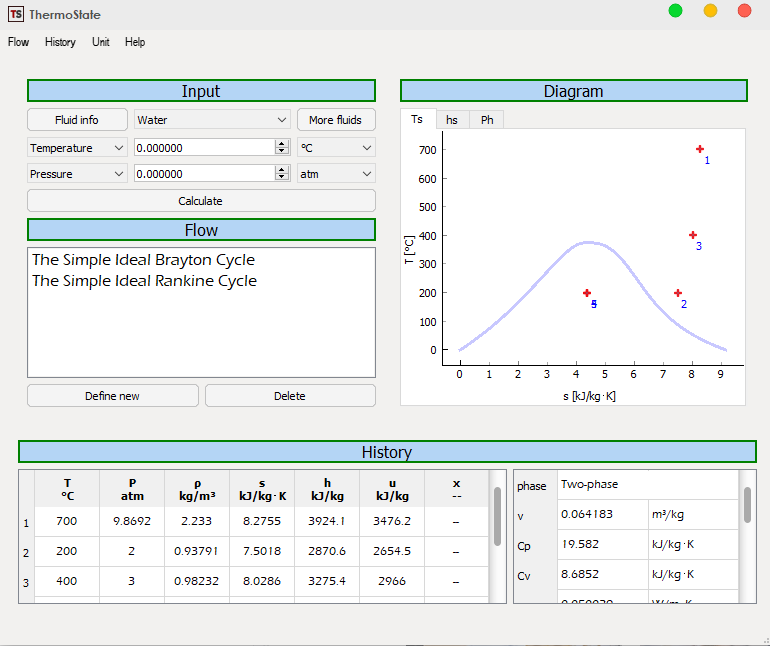

![PDF] Spreadsheet Calculations of Thermodynamic Properties | Semantic Scholar PDF] Spreadsheet Calculations of Thermodynamic Properties | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6af9e71616fdfe0c904c310c6eb020d9e6eb417d/2-Figure1-1.png)

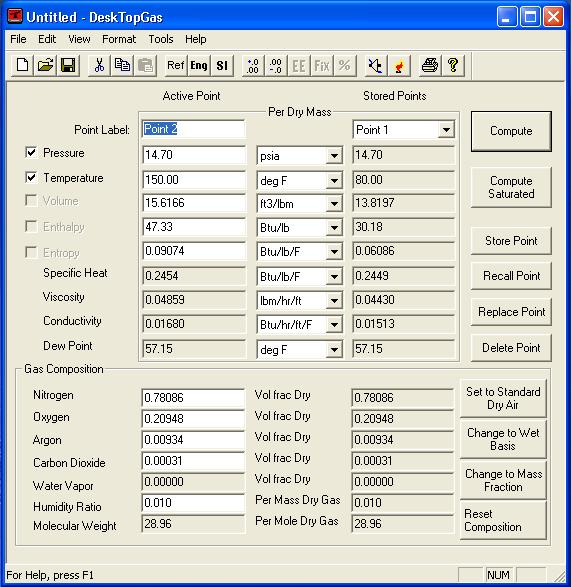

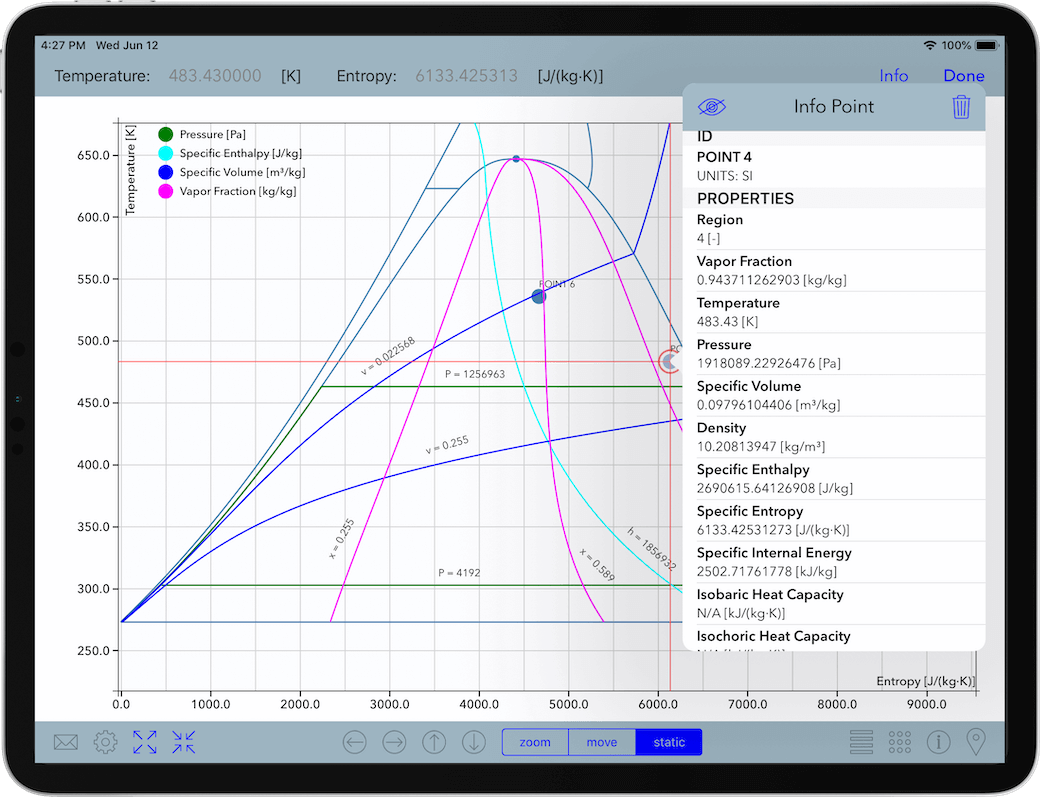
![PDF] Spreadsheet Calculations of Thermodynamic Properties | Semantic Scholar PDF] Spreadsheet Calculations of Thermodynamic Properties | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6af9e71616fdfe0c904c310c6eb020d9e6eb417d/3-Figure2-1.png)